
Rapid medical diagnostic and testing products company
OraSure Technologies Inc. (NASDAQ: OSUR) stock has been a rollercoaster ride this year but is still outperforming the benchmark
S&P 500 index (NYSEARCA: SPY). The medical testing company saw shares collapse (with everyone else) in February during the pandemic sell-off but snapped back hard from the March lows to transcend new multi-year highs. Sentiment in OraSure shares improved significantly as demand for its COVID-19 testing products accelerated in the race towards
COVID-19 vaccine approvals. With the anticipation of two potential FDA emergency use approvals for their antibody and antigen self-tests launching in Q4 2020, prudent investors may want to monitor opportunistic pullback levels for exposure.
Q2 FY 2020 Earnings Release
On Aug. 5, 2020, OraSure released its fiscal second-quarter 2020 results for the quarter ending June 2020. The Company reported an earnings-per-share (EPS) loss of (-$0.16) excluding non-recurring items versus consensus analyst estimates for a loss of (-$0.08), missing estimates by (-$0.08). Revenues fell (-24.5%) year-over-year (YoY) to $29.3 million missing analyst estimates for $32.82 million. Operating loss was (-$9.4) million for the quarter compared to operating income of $5.3 million in Q2 2019. Net product sales and revenues dropped due to lower sales of genomics products and domestic HIV test products as a result of the COVID-19 pandemic. This was partially offset by higher lab service revenues and product sales of COVID-19 testing. International sales of the HIV Self-Test fell (-28%) due to shipping delays but still show robust double digit year-to-date (YTD) growth.
The DNA Genotek business great 4% YoY which included $8.5 million in sales of COVID-19 oral fluid collection devices. Laboratory services nearly doubled YoY to $2.2 million due to COVID-19 testing services. The Company pulled any financial guidance due to uncertainty with COVID-19 but did note that while non-COVID-19 related revenues would be down in 2020, they should be well offset by the surge from COVID-19 related revenues. Most of this will be coming from Molecular solutions business unit. The Company ended the quarter with $265.8 million in cash which includes $95 million net proceeds from a June equity offering.
Conference Call Takeaways
CEO of OraSure, Steven Tang, reiterated his confidence in the FDA approval for the OraSure antigen and antibody tests for COVID-19 under development. The Company expects a Q4 2020 launch of its OraQuick Coronavirus Rapid Antigen Self-Test and the OraSure SARS-CoV-2 oral fluid antibody test. Both tests would detect active COVID-19 infection at the point of collection with no instrumentation needed to interpret results nor need to transport samples to a lab. While the test would initially be used for symptomatic users, eventually asymptomatic users should also be able to use them. The Company has been investing in capacity expansion in parallel with clinicals studies to meet capacity demands upon FDA approval.
The biggest hurdle would be the production constraints after an emergency use FDA approval. The demand could surge to upwards of 30 million tests per week for rapid tests as per a study by The Rockefeller Foundation. The Company anticipates to double total test manufacturing capacity for three product lines being used for COVID-19 molecular sample collection to 70 million tests annually by Q2 and 80 million annually by Q3 of 2021. The Company plans to hire 150 new workers to support capacity expansion efforts.
FDA EUA Approval
On Sept 24, 2020, the FDA approved emergency use authorization (EUA) for a COVID-19 test developed by Quadrant Biosciences which includes the use of ORAcollect-RNA device for collecting saliva swab samples. This is the sixth COVID-19 test using OraSure’s same collection devices and first to exclusively use a saliva-only collection method. Prudent investors looking to gain exposure in the rapid test demand surge into the winter season may consider opportunistic pullback levels.
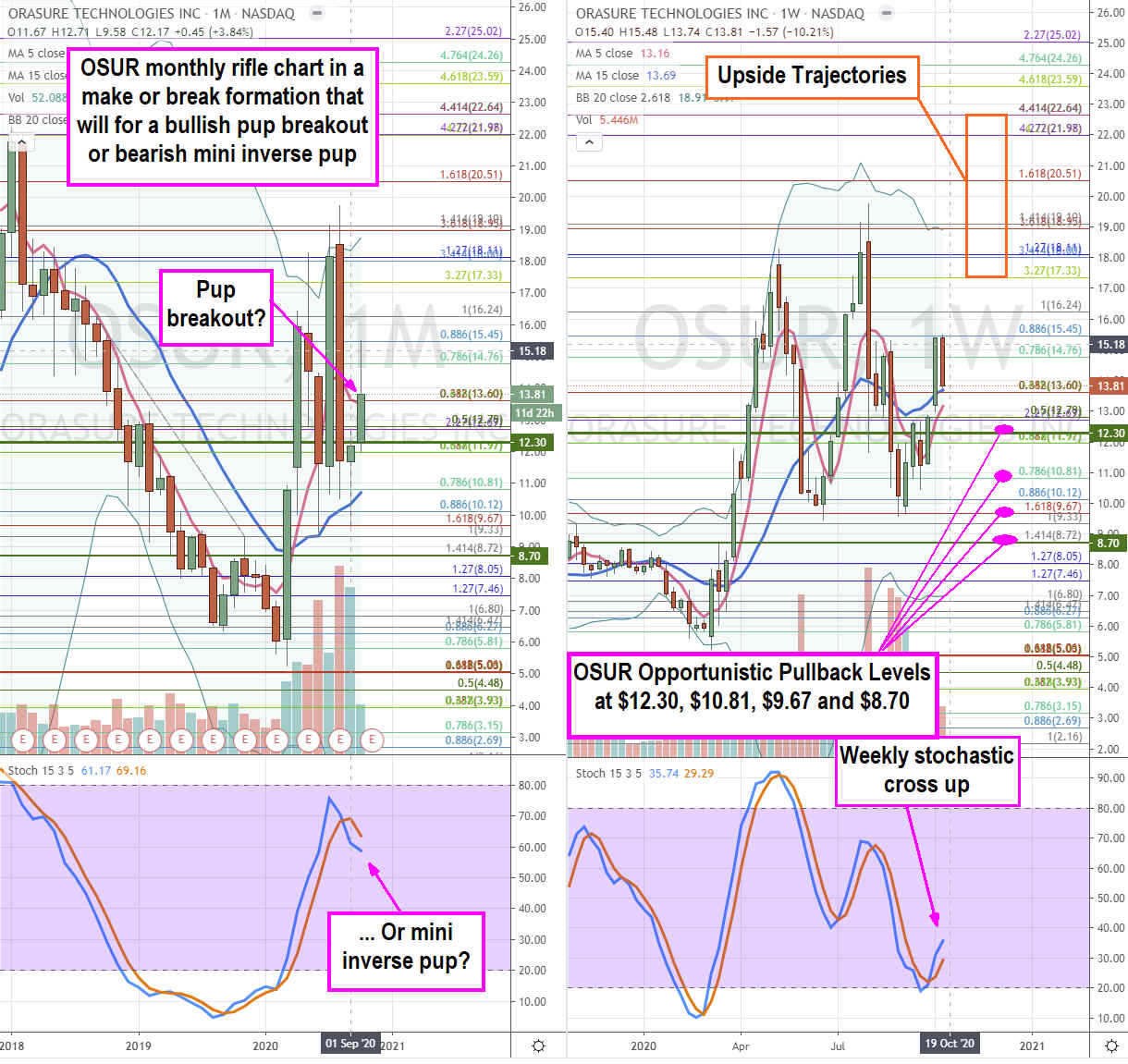
OSUR Opportunistic Pullback Levels
Using the rifle charts on the weekly and daily time frames provides a broader view of the landscape for OSUR stock. The monthly rifle chart is in a make or break situation where the moving averages (MA) could for a pup breakout if the stochastic crosses up or a breakdown is the stochastic forms a mini inverse pup as the 5-period moving average (MA) crossovers down through the 15-period MA. The weekly stochastic has just crossed up but the 5-period MA is still rising at $13.16 after triggering full the weekly market structure low (MSL) above $12.30. The 15-period MA sits at the $13.50 Fibonacci (fib) level. Prudent investors can look for opportunistic pullback levels at the $12.30 weekly MSL/fib, $10.81 fib, $9.67 fib and the $8.70 fib. The upside trajectories range from $17.33 to $22.64 likely requiring the anticipated FDA EUA approval for its antigen and antibody COVID-19 rapid tests. If shares spike ahead of an approval, its best to lock profits as a sell-the-news reaction could form on the actual news.
Companies in This Article: